Quality Assurance
Statgraphics contains several procedures designed to help quality analysts assure that their products meet established standards. In addition to procedures for capability analysis, which are listed on a separate page, this page describes several important quality assurance procedures.
| Methodology |
| Calibration Models |
| Pareto Analysis |
| Stability Studies |
Calibration Models
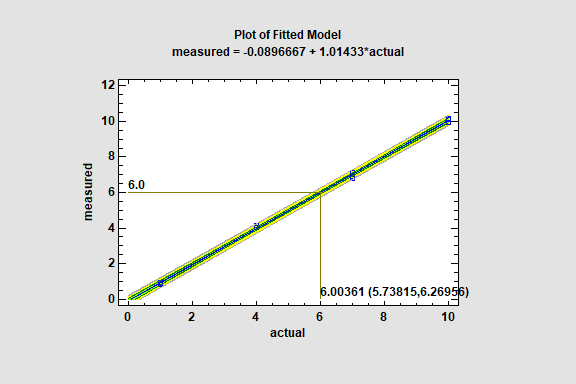
The Calibration Models procedure is designed to construct a statistical model describing the relationship between 2 variables, X and Y, where the intent of the model-building is to construct an equation that can be used to predict X given Y. In a typical application, X represents the true value of some important quantity, while Y is the measured value. Initially, a set of samples with known X values are used to calibrate the model. Later, when samples with unknown X values are measured, the fitted model is used to make an inverse prediction of X from the measured values Y.
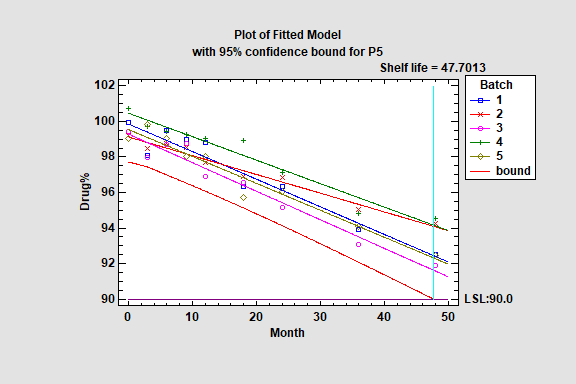
Stability Study
Stability studies are commonly used by pharmaceutical companies to estimate the rate of drug degradation and to establish shelf life. Measurements are typically made on samples from multiple batches at different points in time. Of primary interest is estimating the time at which the lower prediction limit from the degradation model crosses the lower specification limits for the drug.
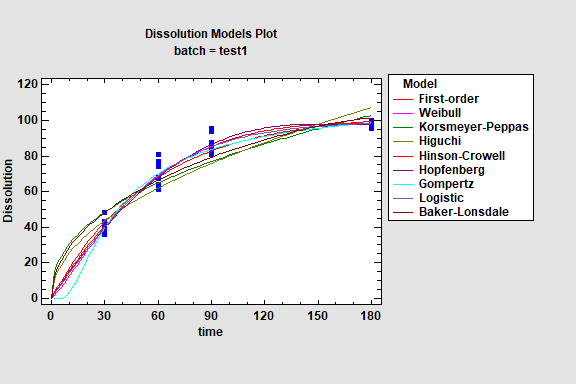
Dissolution Analysis is used to fit dissolution profiles for pharmaceuticals and other types of products. 10 different model types are available which can be examined to select an appropriate model form. Statgraphics contains 3 procedures for analyzing dissolution profiles:
1. Dissolution Model Fitting - identifies an appropriate model for a particular product.
2. Dissolution Model Dependent Comparisons - compares dissolution profiles based on selected dissolution models.
3. Dissolution Model Free Comparisons - used to compare dissolution profiles for pharmaceuticals by calculating similarity statistics between test profiles and a reference profile to determine whether or not those profiles are similar. Does not require fitting a specific dissolution model.
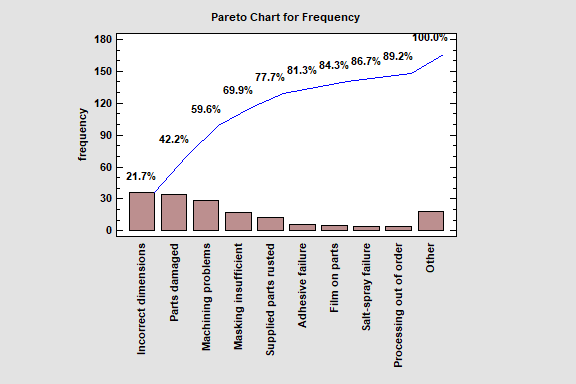
Pareto Analysis is a statistical procedure that seeks to discover from an analysis of defect reports or customer complaints which “vital few” causes are responsible for most of the reported problems. The old adage states that 80% of reported problems can usually be traced to 20% of the various underlying causes. By concentrating one’s efforts on rectifying the vital 20%, you can have the greatest immediate impact on product quality.
Statgraphics supports two basic types of Pareto analysis: weighted and unweighted. In an unweighted analysis, only the frequency of each problem is considered in determining the vital few causes. In a weighted analysis, the cost or impact of each problem is also considered.